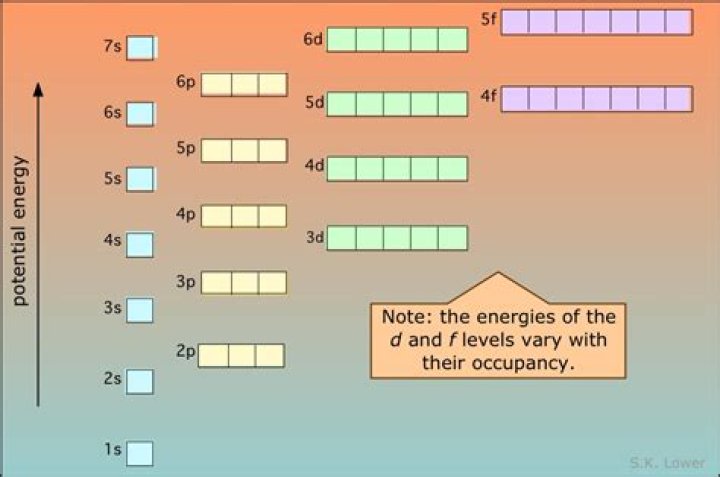
Orbital energies are not physical properties. They are constructs that arise from our approximate approach to a true multi-electron wavefunction using products of single-electron wavefunctions called atomic orbitals.
What determines orbital energy?
Thus, the energy of orbitals depends upon the values of both the principal quantum number (n) and the azimuthal quantum number (l). Hence, the lower the value of (n + l) for an orbital, the lower is its energy.
What is an orbital in physics?
orbital, in chemistry and physics, a mathematical expression, called a wave function, that describes properties characteristic of no more than two electrons in the vicinity of an atomic nucleus or of a system of nuclei as in a molecule. … An s orbital is spherical with its centre at the nucleus.
What is an orbital simple definition?
plural orbitals. Definition of orbital (Entry 3 of 3) physics : a mathematically described region around a nucleus in an atom or molecule that may contain zero, one, or two electrons Electrons arrange themselves in cloudlike regions around the nucleus called orbitals.Do orbitals have same energy?
8 note that the orbital energies depend on only the principal quantum number n. Consequently, the energies of the 2s and 2p orbitals of hydrogen are the same; the energies of the 3s, 3p, and 3d orbitals are the same; and so forth.
Which orbitals have the highest energy?
In all the chemistry of the transition elements, the 4s orbital behaves as the outermost, highest energy orbital.
What is orbital energy astronomy?
For an elliptic orbit the specific orbital energy is the negative of the additional energy required to accelerate a mass of one kilogram to escape velocity (parabolic orbit). For a hyperbolic orbit, it is equal to the excess energy compared to that of a parabolic orbit.
Is orbital energy the same as ionization energy?
Ionization energy depends on orbital energy, which depends on the type of orbital and the effective nuclear charge. … As you go across the periodic table, usually the type of orbital is the same, and the effective nuclear charge increases, making the orbital more stable, so ionization energy increases.What does orbital energy group do?
Orbital Energy Group, Inc. is a publicly traded company. The Company specializes in the acquisition and development of innovative companies to create a diversified energy infrastructure services platform.
What best describes an orbital?The answer is (a) A space in an atom where an electron is most likely to be found.
Article first time published onWhat are examples of orbit?
The definition of an orbit is a circular shape, the rotation of one full circle or a range of experience. The path the earth takes around the sun is an example of the earth’s orbit. The 365 days it takes the earth to get around the sun is an example of the time it takes for a complete orbit or full circle around.
What is an orbit in chemistry?
An orbit in chemistry is the fixed path on which electron moves or revolves around the atom’s nucleus. Furthermore, an orbit is a simple planar representation of a particular electron.
What are the 7 orbitals?
The shape of the seven 7f orbitals (cubic set). From left to right: (top row) 7fy 3, 7fz 3, 7fx 3, (middle row) 7fy(z 2-x 2), 7fz(x 2-y 2), and 7fx(z 2-y 2) (bottom row) 7fxyz. For each, the green zones are where the wave functions have positive values and the white zones denote negative values.
Do electrons orbit?
The electrons do not orbit the nucleus in the manner of a planet orbiting the sun, but instead exist as standing waves. Thus the lowest possible energy an electron can take is similar to the fundamental frequency of a wave on a string.
What is nodal plane BYJU's?
Nodal planes are defined as the planes of zero probability region to find the electron. The number of planes is equal to l. … The density of the dots represents the probability density of finding the electron in that region.
Why does the 1s orbital have the lowest energy?
The Is orbital is spherical in the sense that the electron density varies with distance from the nucleus but not with direction. … An electron in a 1s orbital is of lower energy than one in a 2s orbital because it spends more of its time close to the atomic nucleus.
What are orbitals with the same energy called?
Orbitals that have the same or identical energy levels are referred to as degenerate.
How many orbitals are in the 6th energy level?
The quantum number n represents the shell number, and n = 6 represents the 6th shell (and sixth period on the periodic table). The quantum number l represents the subshell number, and l = 3 represents f-orbitals, so there will be 7 orbitals in a 6f subshell. But only one orbital in 6f subshell will have an ml =-2.
What is electron energy?
The energy of an electron is of the same order of magnitude (is in the same range) as the energy of light. The lines in the spectrum of an element represent changes in the energy of electrons within the atoms of that element. … The energy of an electron depends on its location with respect to the nucleus of an atom.
What is the energy of Nth orbit?
The energy of the electron of hydrogen atom in its nth orbit is given by En=−n213. 6 electron volt(eV).
Does orbiting require energy?
There is no need for energy to be put be into the system to maintain the orbit. Gravitational potential energy is just a different way to describe the work done by gravity.
What is the lowest energy orbital?
The lowest energy sublevel is always the 1s sublevel, which consists of one orbital. The single electron of the hydrogen atom will occupy the 1s orbital when the atom is in its ground state.
What is the difference between orbitals and energy levels?
The main difference between orbitals and energy levels is that orbitals show the most probable pathway of an electron that is in motion around the nucleus whereas energy levels show the relative locations of orbitals according to the amount of energy they possess.
What energy level has the lowest energy?
These zones are known as energy levels (or sometimes called electron shells). At the lowest energy level, the one closest to the atomic center, there is a single 1s orbital that can hold 2 electrons.
Is orbital energy stock a buy?
Orbital Energy Group has received a consensus rating of Buy. The company’s average rating score is 3.00, and is based on 3 buy ratings, no hold ratings, and no sell ratings.
What is the potential energy of a satellite?
Ug = −Gm1m2r
Who owns OEG stock?
StockholderStakeShares bought / soldThe Vanguard Group, Inc.3.33%+313,500Empery Asset Management LP2.78%+1,851,852AWM Investment Co., Inc.1.90%+1,265,669BlackRock Fund Advisors1.07%+104,170
What do you mean by ionization energy?
ionization energy, also called ionization potential, in chemistry and physics, the amount of energy required to remove an electron from an isolated atom or molecule. … Among the chemical elements of any period, removal of an electron is hardest for the noble gases and easiest for the alkali metals.
Which element has highest ionization energy?
Thus, helium has the largest first ionization energy, while francium has one of the lowest.
What is the ionization energy of lithium?
ElementElectron ConfigurationFirst Ionization Energy IE1Lithium (Li)[He]2s1520 kJ/molBeryllium (Be)[He]2s2899 kJ/molBoron (B)[He]2s22p1801 kJ/molCarbon (C)[He]2s22p21086 kJ/mol
How many electrons can an orbital hold?
Orbital: A region of space within an atom where an electron in a given subshell can be found. Any orbital can hold a maximum of 2 electrons with opposite spin. The first shell has one 1s orbital and holds 2 electrons. The second shell holds 8 electrons; 2 in a 2s orbital and 6 in three 2p orbitals.