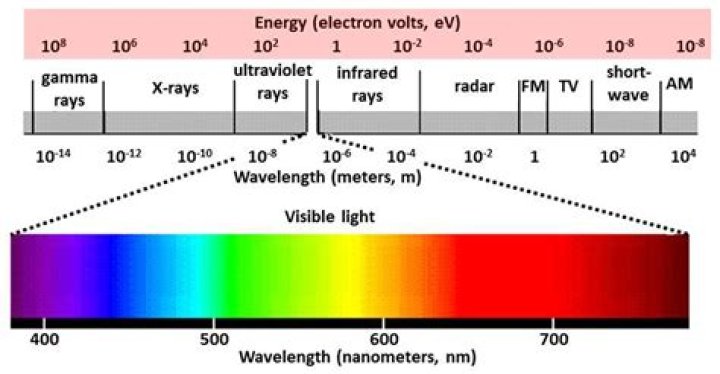
Gamma rays are the type of electromagnetic radiation with the highest amount of energy. They have the shortest wavelength, and therefore the highest frequency.
Which electromagnetic radiation has the highest energy?
Gamma rays have the highest energies and shortest wavelengths on the electromagnetic spectrum.
Which of these has the highest energy?
Answer: (c) Gamma rays Therefore, in the electromagnetic spectrum, gamma rays have the highest energy, and long radio waves the lowest.
Which type of electromagnetic radiation is lowest in energy?
Radio waves have photons with the lowest energies. Microwaves have a little more energy than radio waves. Infrared has still more, followed by visible, ultraviolet, X-rays and gamma rays.What has the most energy?
Fat provides the most energy of all the macronutrients, at a whopping 9 calories per gram.
Which type of radiation has the most energy how or where is this radiation formed?
The different types of radiation are defined by the the amount of energy found in the photons. Radio waves have photons with low energies, microwave photons have a little more energy than radio waves, infrared photons have still more, then visible, ultraviolet, X-rays, and, the most energetic of all, gamma-rays.
What type of radiation has the highest wavelength?
Radio waves, infrared rays, visible light, ultraviolet rays, X-rays, and gamma rays are all types of electromagnetic radiation. Radio waves have the longest wavelength, and gamma rays have the shortest wavelength.
Which of the following orbitals has the highest energy?
The order of the electron orbital energy levels, starting from least to greatest, is as follows: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p.Which of the following photons has the greatest energy?
Gamma rays, a form of nuclear and cosmic EM radiation, can have the highest frequencies and, hence, the highest photon energies in the EM spectrum.
Which of the following electrons have highest energy?Valence electrons are the highest energy electrons in an atom and are therefore the most reactive. While inner electrons (those not in the valence shell) typically don’t participate in chemical bonding and reactions, valence electrons can be gained, lost, or shared to form chemical bonds.
Article first time published onWhich Subshell among the following has the highest energy?
3p = 3 + 1 = 4 The highest value of (n + l) is of 3d so the shell will have the highest energy.
How do you know which wave has the highest energy?
(b) The wave with the shortest wavelength has the greatest number of wavelengths per unit time (i.e., the highest frequency). If two waves have the same frequency and speed, the one with the greater amplitude has the higher energy.
What macromolecule gives the most energy?
There are four classes of biological molecules: fats, carbohydrates, proteins, and nucleic acids. Of these, fats produce the most energy per gram at a whopping nine calories per gram. Carbohydrates and proteins produce less than half of this, at only four calories per gram.
Which of the following electromagnetic radiation have greater frequency?
Gamma rays have the highest frequency in the electromagnetic spectrum.
Which of the following has the highest frequency?
The frequency of Gamma rays is the highest.
What is the highest frequency?
Gamma rays have the shortest wavelengths and highest frequencies of all electromagnetic waves. Gamma rays have more energy than any other electromagnetic waves, because of their extremely high frequencies.
Which particle has highest energy?
Gamma rays have the highest-known electromagnetic energy — those with energies more than a billion times higher than a photon of visible light have been observed by the Fermi Gamma-ray Space Telescope and other ground-based observatories.
What type of radiation has the highest quality factor?
Type of RadiationRadQ FactorBeta Particles11Thermal Neutrons15Fast Neutrons110Alpha Particles120
Which photon has the greatest energy quizlet?
Gamma rays have the most energy per photon of light. The electromagnetic spectrum of light can also be arranged in terms of wavelengths.
In which Subshell is the highest energy electron of tin?
- This electron is located on the fifth energy level, in the p-subshell, in the 5px orbital, and has spin-up.
- This electron is located on the fifth energy level, in the p-subshell, in the 5py orbital, and has spin-up.
Which is higher in energy 4f or 5s?
Even though 5s orbitals have a higher principal quantum number than 4d orbitals, (n = 5 compared to n = 4), they’re actually lower in energy. As a result, 5s orbitals are always filled before 4d orbitals. … 5s, 5p, and 6s orbitals are all lower than 4f orbitals.
Which of the following orbitals has the highest energy according to the Aufbau principle?
The principle formulated by the Danish physicist Niels Bohr about 1920. The 4p orbital has the highest energy capacity among the other orbitals given in the option.
Why do valence electrons have the highest energy?
Pushing the electron up and away from the nucleus increases its potential energy. Because valence electrons are farther from the nucleus than other electrons, they therefore have more potential energy.
Why do valence electrons have the most energy?
So a large amount of energy is required to liberate an electron from an inner most shell rather than an electron from the outermost shell. This is why we say that the electron in the outermost shell has a higher (potential) energy than the inner most shells.
Which of the following electrons has highest energy 25 2p 3p?
electron of 3p orbital has higher energy then 1s, 2s, 2p.
Which shell of argon atom has the highest energy?
K-shell and L-shell have maximum capacity of 2 and 8 electrons respectively. M-shell has a capacity of 18 electrons. Since, it is the outermost orbit and there is a rule that ‘the maximum number of electrons that can be accommodated in the outermost orbit is 8’. In the present case, M-shell is also complete.
Why do gamma rays have the highest energy?
Electromagnetic waves vary in their wavelengths and frequencies. Higher-frequency waves have more energy. Of all electromagnetic waves, gamma rays have the shortest wavelengths and highest frequencies. Because of their very high frequencies, gamma rays have more energy than any other electromagnetic waves.
Do high frequency waves have more energy?
Wave Frequency and Energy The frequency of a wave is the same as the frequency of the vibrations that caused the wave. … This takes more energy, so a higher-frequency wave has more energy than a lower-frequency wave with the same amplitude.
What macromolecule gives us energy?
Carbohydrates provide energy to the body, particularly through glucose, a simple sugar that is a component of starch and an ingredient in many staple foods. Carbohydrates also have other important functions in humans, animals, and plants.