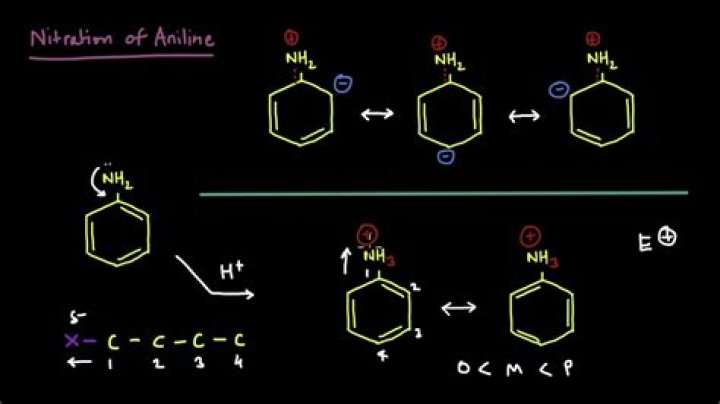
Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitro group, NO2. … Some of the nitrobenzene formed reacts with the nitrating mixture of concentrated acids. Notice that the new nitro group goes into the 3 position on the ring.
What is nitration reaction explain with suitable example?
Benzene reacts with nitric acid and sulphuric acid to form nitrobenzene. … It is an example of electrophilic aromatic substitution reaction. One hydrogen atom of benzene ring is replaced with nitro group. Nitric acid reacts with sulphuric acid to form nitronium ions.
What reagent is nitration?
The key reagent for nitration is nitric acid, HNO3. By itself, nitric acid is a relatively slow-acting electrophile, especially in the presence of a poor nucleophile such as benzene. [Note – in the case of phenol and other aromatic rings with strongly activating groups, HNO3 by itself is sufficient for nitration].
What is used in nitration?
In the Nitration process, the strong electrophile used is Nitronium ion. The formation of the Nitronium ion takes place when the nitric acid is protonated using the sulphuric acid.What are the factors affecting nitration?
Rate of these nitration is proportional to the concentration of the added nitric acid and of organic substrate. Nitration in organic solvents (Mixture of nitromethane or acetic acid with nitric acid): • Kinetics of the process depend upon the aromatic compound being nitrated.
What is Electrophile in nitration?
Electrophile used in the nitration of benzene is nitronium ion NO2+.
What do you understand by nitration describe the mechanism of nitration of benzene?
The mechanism for nitration of benzene: Step 1: Nitric acid accepts a proton from sulphuric acid and then dissociates to form nitronium ion. Step 2: The nitronium ion acts as an electrophile in the process which further reacts with benzene to form an arenium ion.
Why h2so4 is used in nitration?
Sulfuric acid is needed in order for a good electrophile to form. Sulfuric acid protonates nitric acid to form the nitronium ion (water molecule is lost). The nitronium ion is a very good electrophile and is open to attack by benzene. Without sulfuric acid the reaction would not occur.What is nitration reaction Class 11?
Nitration is the class of chemical processes that introduces the nitro group into an organic chemical compound. In the case of organic nitrates, the nitrogen is usually bonded with an oxygen atom which further has a bond to a carbon atom. …
Why is nitration important in chemistry?Nitration is used to add nitrogen to a benzene ring, which can be used further in substitution reactions. The nitro group acts as a ring deactivator. Having nitrogen present in a ring is very useful because it can be used as a directing group as well as a masked amino group.
Article first time published onHow does nitration occur?
Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a nitro group, NO2. Benzene is treated with a mixture of concentrated nitric acid and concentrated sulfuric acid at a temperature not exceeding 50°C. The mixture is held at this temperature for about half an hour.
How is nitration used in the real world?
Nitration reactions are notably used for the production of explosives, for example the conversion of toluene to TNT (2,4,6-trinitrotoluene). However, explosives aside, the nitro compounds are of wide importance as chemical intermediates and precursors. … Nitration is used in the first step of TDI production.
What is nitration of phenol?
Nitration of Phenols. When phenol is treated with concentrated nitric acid, the nitration results in the formation of 2, 4, 6-trinitrophenol (commonly called picric acid). Formation of 2, 4, 6-trinitrophenol.
What are the product of nitration of acetanilide?
In this electrophilic aromatic substitution reaction, the acetamido group (−NHCOCH3) directs the nitronium ion (+NO2) to the ortho and para positions of the aromatic ring. 6 Thus, nitration of acetanilide principally produces ortho- and para-nitroacetanilides, with the para compound being the major product.
How is the electrophile generated in the nitration of benzene?
The arenium ion loses a proton and forms nitrobenzene. Hence, the generation of electrophile in nitration of benzene takes place by protonation of nitric acid with the help of sulphuric acid. The arenium ion is the resonance stabilized carbocation which forms during the electrophilic substitution in an aromatic ring.
How does nitric acid behave in nitration?
Nitric acid reacts with proteins to form yellow nitrated products. This reaction is known as the xanthoproteic reaction. This test is carried out by adding concentrated nitric acid to the substance being tested, and then heating the mixture.
Why nitration reaction is exothermic?
In general, nitration reactions are fast and highly exothermic. … A large proportion of nitric acid consumed during aromatic nitration is directed towards the synthesis of aniline derivatives, which are produced by nitration followed by reduction.
What type of reaction is nitration of benzene?
The type of reaction is classified by its rate-determining step. Since this mechanism has a rate-determining step which involves the attack on the nitronium ion which is an electrophile by the benzene ring electrons, therefore nitration of benzene is an electrophilic substitution reaction.
What is Nucleophile in nitration of benzene?
In this reaction, a hydrogen atom on the benzene ring is substituted with a nitro group (-NO2). The typical reagents for the nitration reaction are nitric acid and sulfuric acid: As with other electrophilic aromatic substitution reactions, the benzene ring acts as a nucleophile.
What is aromatic nitration?
Aromatic nitration is a class of industrially important reactions for the synthesis of chemical intermediates with direct relevance to dyes, plastics, and pharmaceuticals. The nitration reagent is a mixture of concentrated nitric and concentrated sulfuric acid (333). Obviously, a more benign route would be desirable.
Is nh3 an electrophile?
As a result ammonia can never act as an electrophile. This is because there is repulsion between the lone pair of electron and other approaching electrons.
What is the electrophile in nitration of methyl benzoate?
Nitric and sulfuric acid react to form the nitronium ion electrophile. Nucleophilic π electrons attack the nitronium ion giving methyl 3-nitrobenzoate.
What is called nitration?
Definition of nitration : the process of adding a nitro group to an organic compound.
What are nucleophiles 11?
A nucleophile is a chemical species which, in relation to a response, gives an electron pair to form a chemical bond. Any molecule, ion or atom that is in some manner deficient in electron can act as an electrophile.
What is chlorination of benzene?
The halogenation of benzene Benzene reacts with chlorine or bromine in the presence of a catalyst, replacing one of the hydrogen atoms on the ring by a chlorine or bromine atom. … It reacts with some of the chlorine or bromine to form iron(III) chloride, FeCl3, or iron(III) bromide, FeBr3.
What is the major product of nitration of toluene?
The nitration of toluene results in ortho and para nitrotoluene isomers. When it is heated it results in dinitrotoluene and trinitrotoluene which is explosive.
Which is most readily nitrated?
Phenol is more easily nitrated than benzene.
Which catalyst is used in nitration?
Nitration is an important chemical reaction widely used in commercial manufacturing of various nitro-aromatics. Concentrated H2SO4 has been currently used as the most effective and efficient catalyst in a large-scale liquid phase nitration process.
What is aromaticity in organic chemistry?
In chemistry, aromaticity is a property of cyclic (ring-shaped), typically planar (flat) structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to other geometric or connective arrangements with the same set of atoms.
What acid makes nitrate?
Nitric acid is used in the production of ammonium nitrate and calcium ammonium nitrate which find applications as fertilizers.
What are the different types of nitration?
Nitration is the most studied and researched organic reactions. Both the aromatic and aliphatic compounds can be nitrated by different methods such as heterolytic (electrophilic and nucleophilic) and the radical nitrations. Aromatic nitration is the most frequent, and the Aliphatic is a free radical electrophilic.