Aluminum is the easiest to oxidize due to its position on the periodic table as it has 3 valence electrons.
What metal is easily oxidized in rocks?
Generally speaking, metals including sodium, magnesium, and iron are easily oxidized.
Is aluminum easily oxidized?
Aluminum oxidation happens faster than that of steel, because aluminum has a really strong affinity for oxygen. Rather than flaking though, aluminum oxide just forms a hard, whitish-colored surface skin. When all the aluminum atoms have bonded with oxygen the oxidation process stops.
Is copper easily oxidized?
Copper is a versatile metal used in thousands of everyday products. It oxidizes readily to form a distinctive coating known as patina. The patina gives the Statue of Liberty its characteristic appearance, but the oxidation of copper can also cause undesirable effects under some circumstances.Which metals oxidize the most?
The order of some common metals in the electromotive series, starting with the most easily oxidized, is: lithium, potassium, calcium, sodium, magnesium, aluminum, zinc, chromium, iron, cobalt, nickel, lead, hydrogen, copper, mercury, silver, platinum, and gold.
Is iron easily oxidized?
In its natural state, however, iron is highly susceptible to rusting. Over time, the versatile metal will become corrode, resulting in the formation of iron oxide, which is more commonly known as rust.
Is Fe easily oxidized?
Fe2+ is easy to oxidize to Fe3+ because removing the electron results in a half filled d subshell. Fe2+ is easy to oxidize to Fe3+ because ions with an odd charge are most stable for atoms with an even atomic number.
Does bronze oxidize?
Copper, Bronze and Brass Copper oxidizes over time to form a green patina, which actually protects the metal from further corrosion. Bronze is a mixture of copper and tin, along with small amounts of other elements, and is naturally much more resistant to corrosion than copper.Is silver easily oxidized?
Silver NPs oxidize easily on the surface. Even though the plasmonic properties are influenced, it can be still used for any plasmonic application.
Is AG more easily oxidized than Cu?No. Since Cu is more active than Ag , it is disfavored for this reaction to occur, as copper would more rather be oxidized than silver would (and thus copper would less rather be reduced than silver would).
Article first time published onDoes stainless steel oxidize?
Stainless steel is low maintenance, and its resistance to oxidation and staining makes it an ideal material for many applications.
Which metal can be oxidized Cu?
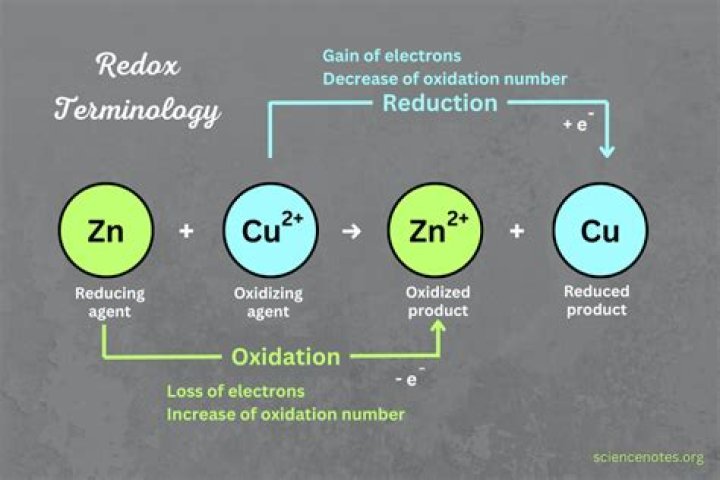
In the first reaction, the copper ion is able to oxidize the zinc metal. However, in the second reaction, the zinc ion is not able to oxidize the copper metal. Zinc is a better reducing agent than copper.
Does steel corrode?
The corrosion products of steel are oxide particles and have a distinctive brown/red (rust) color. … Steel corrodes naturally when exposed to the atmosphere, but the corrosion process accelerates when electrochemical corrosion cells are active on the surface.
Which of the following is the easiest to oxidize?
Lithium is an alkali metal. Alkalis are easily oxidized.
Why are some metals easily oxidized?
The most common kinds of corrosion result from electrochemical reactions. General corrosion occurs when most or all of the atoms on the same metal surface are oxidized, damaging the entire surface. Most metals are easily oxidized: they tend to lose electrons to oxygen (and other substances) in the air or in water.
Which species is most easily reduced?
Use the reduction potential chart: nonmetals are at the top and are most easily reduced. Metals are at the bottom and are most easily oxidized. Lithium is at the bottom of the chart—it’s the most easily oxidized of all.
Is H more active than CU?
Explanation: For example, all alkali metals, sodium, potassium, lithium, francium, etc. are more reactive than hydrogen. … However, metals like gold, copper, and platinum are less reactive than hydrogen.
Which is easier to oxidize copper or silver?
What about copper versus silver? Copper still has a higher electronegativity than silver, but copper metal is more easily oxidized.
Is Cu oxidized or reduced?
Copper is oxidized from 0 to +2, so Cu is the reducing agent.
Is aluminum easier to oxidize than copper?
Corrosion through exposure to oxygen Aluminium oxidizes readily when exposed to air. … Copper also oxidizes when exposed to air, but to a much lesser extent.
Which elements are hardest to oxidize?
Best place to live, hardest to oxidize: the noble metals, silver, mercury, platinum, gold.
Which of the following is most easily oxidized apex?
Easily oxidized elements readily release electrons. The loss of an electron allows these elements to form a stable valence electron configuration. In the context of this question, lithium is the most willing to release an electron, so it is the most easily oxidized.
Does gold oxidize?
Gold is one of the least reactive elements on the Periodic Table. It doesn’t react with oxygen, so it never rusts or corrodes. Gold is unaffected by air, water, alkalis and all acids except aqua regia (a mixture of hydrochloric acid and nitric acid) which can dissolve gold.
Which metal is easiest to reduce?
In the table provided, the most easily reduced element is Li and the most easily oxidized is iron.
Does platinum oxidize?
Platinum is a silver-white metal — it was once known as “white gold.” It is extremely resistant to tarnishing and corrosion (which makes it known as a “noble metal”) and is very soft and malleable, making it easy to shape; ductile, making it easy to stretch into wire; and unreactive, which means it doesn’t oxidize and …
Which metal is the most resistant to corrosion?
1. Stainless steel. Stainless steel alloys are renowned for the corrosion-resistance, ductility, and high strength. Corrosion resistant qualities in stainless steels are directly tied to their chromium and nickel content — more of these elements correlate with increased resistance.
What metal oxidizes green?
When copper reacts with oxygen, it oxidizes and generates a greenish-blue layer that protects the metal from further corrosion. Any metal that contains a high amount of copper can turn green. Actually, this green layer has a number of benefits, and it’s often used intentionally.
What is oxidized copper?
Oxidized copper is a specific type of corrosion that is produced during a three-step process where copper oxidizes to copper oxide, then to cuprous or cupric sulfide, and finally to copper carbonate. It results in a green-colored copper layer or patina that forms over time.
Is silver easily reduced?
In general, the ions of very late transition metals — those towards the right-hand end of the transition metal block, such as copper, silver and gold — have high reduction potentials. In other words, their ions are easily reduced.
Is Ag or Cu reduced?
Cu(s)2 Ag(s)stronger reducing agentweaker reducing agent
Does gold oxidize iron?
Gold is a pure metal which doesn’t rust because it doesn’t contain iron. “Gold is the most non-reactive of all metals and is benign in all natural and industrial environments.