The reaction, also called the Goldschmidt process, is used for thermite welding, often used to join railway tracks. Thermites have also been used in metal refining, disabling munitions, and in incendiary weapons. Some thermite-like mixtures are used as pyrotechnic initiators in fireworks.
Is it legal to use thermite?
Yes they can. In thermite there is a metal which is the fuel and a metal oxide which provides the oxygen. For example, mix of aluminium or magnesium and iron oxide is used in thermite welding.
What can thermite burn?
Thermite, a mixture of metal powder and metal oxide, is the hottest burning man-made substance in the world. It burns at temperatures of more than 2,200C, enough to burn through steel or asphalt.
Can you cook with thermite?
The thermite reaction quickly formed liquid iron which dripped down out of the pot, and into a ceramic pan. I put a beef kebab directly onto the liquid iron, which cooked the food in under a minute. It was delicious!Can a lighter ignite thermite?
Reaction and ignition It is difficult to ignite thermite. … A mixture of potassium permanganate and glycerin can be used to ignite thermite. It can be ignited by a flint lighter or a child’s sparkler. It can be mixed with sulfur to make thermate, which makes it easier to melt the iron.
Can you make thermite from an Etch A Sketch?
Etch-a-Sketch Thermite Reaction Mix together the iron oxide and aluminum. Use a sparkler to light the mixture. Move away from the reaction and let it burn to completion before cleaning it up. Once it is cool, you can pick up the molten metal and examine it.
Does thermite burn underwater?
Thermite, a mixture of rust and aluminum. YouTube/TheBackyardScientist This flowerpot full of red powder looks pretty innocuous. But when ignited with a strip of magnesium and a blowtorch, it yields a molten metal so hot it keeps burning underwater.
How do you ignite thermite?
- First, the Mg ribbon can be ignited using a bunsen burner flame. The ribbon can then be dropped into the flower pot, igniting the thermite.
- Second, the Mg ribbon can be inserted into the powder like a fuse, then lit with the bunsen burner.
Are sparklers made of thermite?
ARE SPARKLERS THERMITE? The sticks of the sparklers are coated with a pyrotechnic composition known as ‘Thermite’, which is responsible to act as a fuel in the burning process. So yes, sparklers are thermite-positive. Apart from that, it is also responsible for the redox reaction during the sparkling effect.
Why does thermite and ice explode?Some experts suggest that the reaction releases a huge amount of energy, causing the ice to turn into a vapour. This vapour condenses around the thermite particles, forming a steam-thermite aerosol. As the mixture continues to react, the particles are thrown up into the air in a cloud of thermite, causing an explosion.
Article first time published onDoes thermite explode?
Thermite is a pyrotechnic composition of metal powder, which serves as fuel, and metal oxide. When ignited by heat, thermite undergoes an exothermic reduction-oxidation (redox) reaction. Most varieties are not explosive, but can create brief bursts of heat and high temperature in a small area.
Does thermite burn through concrete?
Oh yes, thermite can burn through concrete. Thermite burns at about 5000 degrees F., or half the temperature at the surface of the sun.
Can thermite be lit with a match?
sufficient to ignite Thermite. The Magnesium Ribbon itself can easily be ignited with a small pocket torch or propane torch. Using a disposable lighter or match generally doesn’t work very well due to their lower flame temperature. The best way to start the Thermite reaction is to use Thermite Ignition Mixture.
What are thermite grenades?
Thermite grenades are a type of explosive incendiary ordnance used by the UNSC. The flames from the thermite grenade can burn underwater. Thermite grenades rely on a chemical process between metal powders and oxides to create high temperatures, with no explosion.
How do you stop thermite?
A fire extinguisher should be readily available at all times. Water should not be used to extinguish the reaction, since addition of water to hot iron produces potentially explosive hydrogen gas.
Is thermite hotter than the sun?
The temperature of the thermite reaction is about 3000C, so close to the temperature of the surface of the sun is an exaggeration, it can melt its way through a garden plant pot as in the clip, but when it is used in the rail industry they use more robust reusable equipment.
Can there be fire in space?
Fires can’t start in space itself because there is no oxygen – or indeed anything else – in a vacuum. Yet inside the confines of spacecraft, and freed from gravity, flames behave in strange and beautiful ways. They burn at cooler temperatures, in unfamiliar shapes and are powered by unusual chemistry.
What are the metal shavings in breaking bad?
Aluminum amalgam is a chemical substance formed by a solution of aluminum with mercury. … In Breaking Bad, aluminum amalgam is shown to be usually forgotten in methamphetamine cook sessions (“Box Cutter”).
How did Walter make thermite?
In order to steal the methylamine they need to continue cooking, Walter White hits upon the idea of making thermite to cut through the warehouse lock after seeing some old Etch-a-Sketch like toys on a shelf. Walt empties the aluminum powder from several of the toys and mixes it with powdered iron oxide to create …
What did Walt get thermite from?
In season 1, episode 7, Walt and Jesse need a way to break into a chemical warehouse to steal a barrel of methylamine. Walt takes inspiration from an etch-a-sketch in Jesse’s garage that they crack open to harvest aluminum powder. They use it to produce thermite, which dramatically burns through the warehouse’s lock.
What is the iron oxide?
Iron oxides are chemical compounds composed of iron and oxygen. There are sixteen known iron oxides and oxyhydroxides, the best known of which is rust, a form of iron(III) oxide. … Iron oxides are inexpensive and durable pigments in paints, coatings and colored concretes.
What is the difference between thermite and Thermate?
Thermate is a variation of thermite and is an incendiary pyrotechnic composition that can generate short bursts of very high temperatures focused on a small area for a short period of time. It is used primarily in incendiary grenades.
What is thermite process explain?
noun. a process for reducing metallic oxides using finely divided aluminium powder. The mixture of aluminium and the oxide is ignited, causing the aluminium to be oxidized and the metal oxide to be reduced to the metal. Also called: thermite process.
Why does aluminum react with iron oxide?
Aluminium reacts with iron(III) oxide because it can be considered to be more reactive than iron so will remove oxygen from the iron(III) oxide.
How do you make iron oxide thermite?
The thermite recipe for red iron oxide and aluminum is 3 parts iron oxide red to 1 part aluminum fuel. In pyrotechnic compositions the finer the ingredients and more well mixed they are, generally the easier it is to ignite, and the faster the reaction will progress.
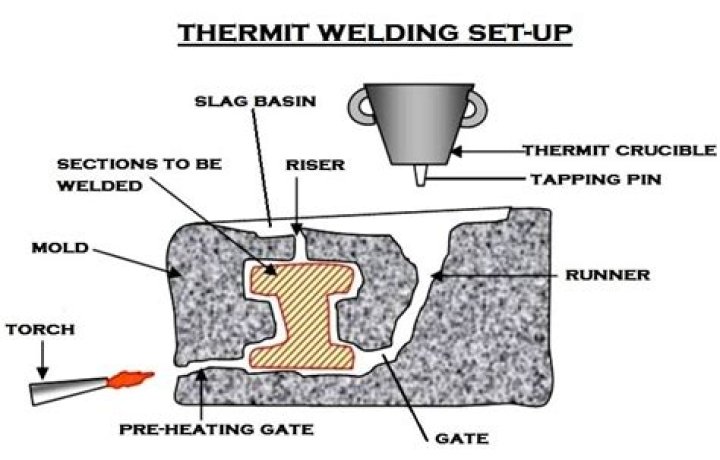
Is thermite used in welding?
Thermite welding is the process of igniting a mix of high energy materials, (also called thermite), that produce a molten metal that is poured between the working pieces of metal to form a welded joint. It was developed by Hans Goldschmidt around 1895. … Thermite welding is widely used to weld railroad rails.
How hot does a thermite grenade burn?
A portion of the thermate mixture is converted to molten iron, which burns at 4,000 degrees Fahrenheit. It will fuze together the metallic parts of any object that it contacts. Thermate is an improved version of thermite, the incendiary agent used in hand grenades during World War II.
What is hot ice?
Sodium acetate or hot ice is an amazing chemical you can prepare yourself from baking soda and vinegar. You can cool a solution of sodium acetate below its melting point and then cause the liquid to crystallize. The crystallization is an exothermic process, so the resulting ice is hot.
What does thermite and ice make?
Heat from the Thermite reaction melts the ice into liquid water, which can now combine with the reacting Thermite. Burning metals like those in Thermite (Iron, Aluminum) react faster with water than with oxygen, so more energy is released.
What happens when you mix fuel metal oxide and metal powder?
A thermite reaction is an exothermic oxidation-reduction reaction similar to the ignition of black powder. The reaction requires a metal oxide and fuel. The fuel in the thermite reaction you produce is aluminum in the foil. Your metal oxide is iron oxide, more commonly known as rust.
What temperature does steel melt at?
Melting PointsMetalsFahrenheit (f)Celsius (c)Silver, Sterling1640893Steel, Carbon2500-28001371-1540Steel, Stainless27501510