OCN^- ONC^- NOC^- OCN^1 ONC^- NOC^- None of these ions are stable according to Lewis theory. All of these compounds are equally stable according to Lewis theory.
How stable is OCN?
The cyanate ion (OCN-) and the fulminate ion (CNO-) share the same three atoms but have vastly different properties. The cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. The resonance structures of the cyanate ion were explored in Example 5.6.
What is the shape of OCN?
Note : $ OC{N^ – } $ has a triple bond between carbon and nitrogen atoms and that is why it can be termed as an ambidentate ligand. This cyanate ion is stable. Its electron pair geometry is tetrahedral and the molecular geometry is trigonal pyramidal.
What makes a Lewis structure most stable?
An atom, molecule, or ion has a formal charge of zero if it has the number of bonds that is typical for that species. Typically, the structure with the most charges on the atoms closest to zero is the more stable Lewis structure.Is OCN 1 polar or nonpolar?
(e) Since the C−O and C≡N C ≡ N bonds have different polarities, the molecule has a non-zero dipole moment and therefore it is polar in nature.
Which of the following ions is most stable?
Tertiary carbonium ions are most stable due to hyperconjugation effect and +I effect of methyl group.
What is the best Lewis structure for seo42?
Recall that SeO42- has an overall –2 formal charge. The structure is a possible Lewis structure for SeO42-. Structure 4 is preferred because the formal charge of the central atom is zero and it has the least atoms with formal charges.
Does OCN have resonance structures?
For the example of OCN–, there are three non-equivalent resonance structures, depending on how the multiple bonds are formed in Step 6 of the Lewis structure drawing procedure. For non-equivalent resonance structures, the bonding and charge distributions are different, so they are in different energy levels.Which bond Below is the weakest?
The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.
Is OCN a resonance?The structure is resonant.
Article first time published onWhich structure is most stable?
- The resonance structures in which all atoms have complete valence shells is more stable. …
- The structures with the least number of formal charges is more stable. …
- The structures with a negative charge on the more electronegative atom will be more stable.
What's the most stable structure?
The most stable structure is one that has the fewest number of, and smallest magnitude of, formal charges and with the negative formal charges on the more electronegative atoms.
Which structure contributes most to the hybrid?
The most stable structures contribute most to the resonance hybrid. They are called the major resonance contributors. In order of importance, a major contributor must have: The most atoms with complete octets.
How many electron groups are there in OCN?
The Lewis structure of OCN– is: Step 4: There are two electron groups around the central atom. Electron groups include lone pairs and atoms around the central atom: for OCN–, there are 0 lone pairs and 2 atoms, or a total of 2 electron groups around C. There are two (2) electron groups around the carbon atom.
Which one of the following is the best structure for the fulminate ion CNO −?
Carbon has the negative charge (-3) while oxygen (which is more electronegative) has a (+1) formal charge. Therefore, the best Lewis structure for fulminate ion is structure B.
Does OCN have isomers?
OCN- and CNO- are isomers.
Which bond is most polar?
Fluorine has highest electronegativity while iodine has least electronegativity among group 17 elements. Thus the electronegativity difference between fluorine and iodine is highest due to which they form the most polar bond.
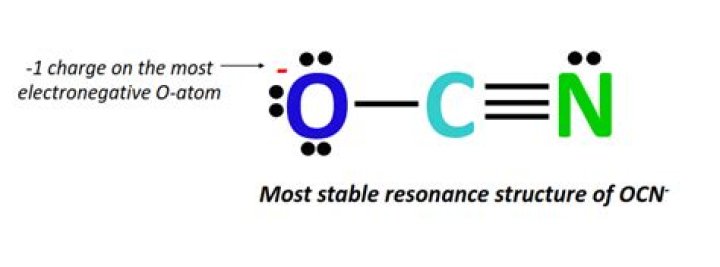
What is the formal charge on the O atom in OCN?
When we calculate the formal charges, we’ll find that the Oxygen has a formal charge of -1, Carbon is 0, and Nitrogen is 0. That makes a lot of sense: we have a -1 up here. The negative charge in our OCN- Lewis structure is on the most electronegative atom. So this looks like a pretty good Lewis structure.
What is the most preferred resonance structure of oxygen is the central atom in the con ion?
ElementNonbonded Electrons(a)H Cl0 6(b)C F0 6(c)P Cl2 6(d)P F0 6
Which is more stable v3+ or v2+?
the outer most electronic configuration of V is 3d^3 4s^2. the stable state of s orbital is o and 2.. … And hence V^+2 is more stable than V^+3.
Which of the following ions is least stable?
Be− is the least stable ion, Be (1s22s2) has stable electronic configuration, addition of electron decreases stability.
Which ion is most stable in aqueous solution nh4+?
NH₄⁺ is the most stable atom. Explanation: The stability of the cations depend on the nature of bonds formed between the central atom and peripheral ions.
Which bond is the strongest bond?
In chemistry, covalent bond is the strongest bond. In such bonding, each of two atoms shares electrons that binds them together. For example, water molecules are bonded together where both hydrogen atoms and oxygen atoms share electrons to form a covalent bond.
Which orbital overlap is strongest?
During the axial overlap of p-p orbitals, the electron density increases around the axis, so the bond formed is the strongest. Therefore, the strongest bond formed is when p-p orbital overlap occurs. Final answer: The correct answer is Option B- 2p and 2p orbitals.
What bonds are strongest to weakest?
The ranking from strongest to weakest bonds is: Covalent bond > ionic bond > hydrogen bond > Van der Waals forces. Complete answer: The order from strongest to weakest bonds is: Covalent bond > ionic bond > hydrogen bond >Van der Waals forces.
Does resonance affect stability?
Resonance stabilization Because resonance allows for delocalization, in which the overall energy of a molecule is lowered since its electrons occupy a greater volume, molecules that experience resonance are more stable than those that do not.
Why does charge separation decrease stability?
This is because in the second Resonating Structure the charges have to be seprated and as both charges are opposite in nature they will attract each other and make the process of seprating them difficult making it unstabilized.
Is there electron delocalization in OCN?
From a delocalization viewpoint, the second electron pair of the C=O. double bond and the nitrogen lone pair both have been delocalized over the entire O-C-N region (lower right). … There are two important structural consequences of this bonding.
Does OCN follow the octet rule?
The cyanate ion (OCN-) has three resonance structures. Each follows the octet rule. (#1) has a oxygen to carbon single bond and a carbon to nitrogen triple bond. … (#3) has both a oxygen to carbon and a carbon to nitrogen double bond.
What is OCN ion?
Cyanate is an anion with the structural formula [O=C=N]−, usually written OCN−. It also refers to any salt containing it, such as ammonium cyanate. … The cyanate ion is an ambidentate ligand, forming complexes with a metal ion in which either the nitrogen or oxygen atom may be the electron-pair donor.
Which one of the following is not a resonance structure of cyanate OCN )?
Lastly talking about the third resonating structure: $ O \equiv C – {N^{2 – }} $ here in this the nitrogen atom is sharing one electron and also there are $ – 1 $ and $ – 2 $ charges already present ten further it will become $ – 3 $ charges on nitrogen atom.