The benzoate ion will then act as a weak base and undergo hydrolysis with water to form benzioic acid and hydroxide ions.
What is the pH of sodium benzoate?
pH% Active Benzoic Acid513.761.5570
Is sodium benzoate a salt or base?
Sodium benzoate is an organic sodium salt resulting from the replacement of the proton from the carboxy group of benzoic acid by a sodium ion. It has a role as an antimicrobial food preservative, a drug allergen, an EC 1.13.
Is benzoate an acid or a base?
Benzoic acid, as its name implies, is an acid. Sodium benzoate is a salt of the conjugate base, the OBz- or benzoate ion. Whenever sodium benzoate dissolves in water, it dissociates into its ions. The benzoate ion then acts as a base toward water, picking up a proton to form the conjugate acid and a hydroxide ion.Why is benzoate a stronger base than formate?
After removal of acidic H, the created conjugate bases formate ion is more stable than benzoate ion since +R destabilizes the benzoate ion and such destabilization is absent by H in formate ion.
What is the solubility of sodium benzoate in water?
Sodium benzoate is highly soluble in water (66 g/100 mL at 20 °C).
Why does sodium benzoate dissolve in water?
The abundant OH- ions will react fully with benzoic acid in acid-base reaction to form water and benzoate ion, which now is soluble in water due to strong ion-dipole forces of attraction.
Is sodium benzoate soluble in oil?
NamesSolubility in water62.69 g/100 mL (0 °C) 62.78 g/100 mL (15 °C) 62.87 g/100 mL (30 °C) 71.11 g/100 mL (100 °C)Solubilitysoluble in liquid ammonia, pyridineIs benzoate soluble in water?
Sodium benzoate is the sodium salt of benzoic acid . It is an aromatic compound denoted by the chemical formula C7H5NaO2 with a molecular weight of 144.11. In its refined form, sodium benzoate is a white, odorless compound that has a sweet, astringent taste, and is soluble in water .
What does sodium benzoate do?In the food industry, sodium benzoate is used to prevent spoilage from harmful bacteria, yeasts, and molds. It also helps maintain freshness in food by helping to slow or prevent changes in color, flavor, PH, and texture. Other foods that commonly include sodium benzoate include: Salad dressings.
Article first time published onIs sodium benzoate a solid liquid or gas?
The Chemistry of Sodium Benzoate In its solid form it is a white, granular or crystalline powder that can be added to food or cosmetics. Other sodium compounds with similar names are sodium borate or borax and sodium carbonate or soda.
Is sodium benzoate polar or nonpolar?
Sodium benzoate is not a molecule. It is a compound made up of two ions, Na+ and C6H5COO-. Benzoic acid consists of molecules. The benzoic acid molecule is polar because the oxygen atoms, which are more electronegative than carbon and hydrogen, are concentrated on one side of the molecules.
Is sodium benzoate in milk?
The major preservatives in dairy products are sodium benzoate, potassium sorbate, and natamycin. The maximum permitted levels for these additives in cheese and yogurt are established according to Iranian national standards.
Is sodium benzoate ionic or covalent?
Sodium Benzoate is a mostly covalent due to the carbon in the benzoate but also it has slightly a ionic bond because of the metal sodium.
What is sodium benzoate in skin care?
Sodium benzoate is used in a wide variety of cosmetics and personal care products where it acts as a corrosion inhibitor, fragrance ingredient, and preservative. As a preservative, sodium benzoate is primarily an anti-fungal agent but also has some effectiveness against bacteria.
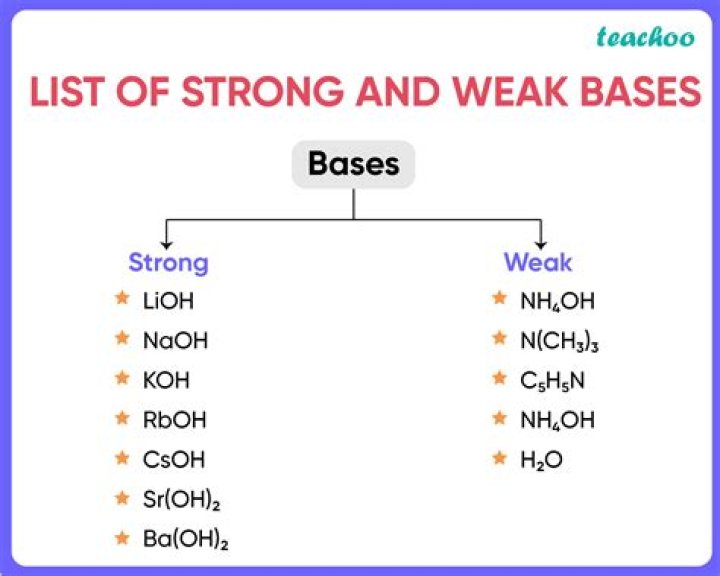
What are some strong bases?
- LiOH (lithium hydroxide)
- NaOH (sodium hydroxide)
- KOH (potassium hydroxide)
- Ca(OH)2 (calcium hydroxide)
- RbOH (rubidium hydroxide)
- Sr(OH)2 (strontium hydroxide)
- CsOH (cesium hydroxide)
- Ba(OH)2 (barium hydroxide)
Which is the strongest base CN or Hcoo?
SInce Ka for formic acid is five orders of magnitude greater than HC≡N , it follows that cyanide ion is a MUCH stronger base than formate.
Are Carboxylates weak bases?
The carboxylate ion has two electronegative oxygens to only one for the alkoxide ion. … The somewhat paradoxical outcome of this is that carboxylic acids are stronger acids than alcohols because carboxylate ions, their conjugate bases, are weaker bases than alkoxides.
Is sodium benzoate soluble in cold water?
Sodium benzoate is the sodium salt of benzoic acid used as a white crystalline or amorphous (without crystal structure) powder, very soluble in water (66 g of sodium benzoate in 100 g of water at 20 °C) but poorly soluble in alcohol.
Is sodium benzoate soluble in ether?
No but sodium benzoate will dissolve in water and benzoic acid won’t. Conversely benzoic acid will dissolve in diethyl ether but not in water. Therefore you can use a water/diethyl ether partition to separate these components in a separatory funnel.
How do you dilute sodium benzoate?
Dissolve in 50 ml of water. Shake continuously for 15 min and Dilute to 100 ml with water.
How sodium benzoate works as a preservative?
Sodium benzoate inhibits the growth of potentially harmful bacteria, mold, and other microbes in food, thus deterring spoilage. … Therefore, it’s commonly used in foods, such as soda, bottled lemon juice, pickles, jelly, salad dressing, soy sauce, and other condiments.
Is sodium benzoate solubility in organic solvents?
Solubility in organic solvents / fat solubility 13.3 g/L.
Is sodium benzoate safe for babies?
Given that children are often consuming vitamin-C containing foods, it’s best to avoid sodium benzoate altogether.
How is sodium benzoate metabolized?
Sodium benzoate is a widely used preservative found in many foods and soft drinks. It is metabolized within mitochondria to produce hippurate, which is then cleared by the kidneys.
What contains sodium benzoate?
Where is Sodium benzoate found? Sodium benzoate is a preservative that can be found in acidic foods such as salad dressings, carbonated drinks, jams, juices, and condiments. It is also found in mouthwashes, silver polishes, cough syrups, soaps, and shampoos.
How does sodium benzoate inhibit bacterial growth?
Sodium benzoate is food preservative that inhibits microbial growth. … incubation time, sodium benzoate concentrations of 2.0 mg/mL increased chromosome break when comparing with the control group (P < 0.05). Sodium benzoate did not cause Ala40Thr (GCG→ACG) in superoxide dismutase gene.
Is sodium benzoate hygroscopic?
Sodium benzoate (CAS No. 532-32-1; C7H5O2Na; benzoic acid, sodium salt [E 211 (EU No. Regulation on Labelling of Foodstuffs)]; molecular weight 144.11) has a melting point above 300 °C. It is very soluble in water (550–630 g/litre at 20 °C) and is hygroscopic at a relative humidity above 50%.
Is benzoate the same as benzene?
No, however, in foods there can be an issue when Sodium Benzoate combines with Ascorbic Acid creating a form of benzene, a known carcinogen. In foods, fats and sugars hinder the formation of benzene and thus is considered safe and usable in foods by FDA. …
What intermolecular forces are in sodium benzoate?
The strongest intermolecular forces between water and sodium benzoate are ion-dipole interactions.
Is sodium benzoate or benzoic acid more polar?
Which is more polar, benzoic acid or sodium benzoate? Sodium Benzoate because it contains Na+ ions which are water soluble, and create a strong dipole moment making the molecule polar, so it dissolves in polar substances such as water.