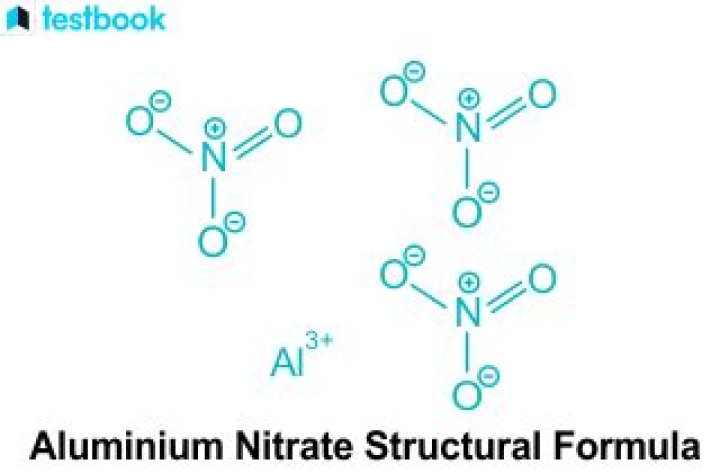
Salts of strong acids and weak bases dissociate and react in water forming H+. When Aluminum Nitrate is dissolved in water, the salt first dissociates in water: … The solution is acidic because Nitric acid is a strong acid while Aluminum is a weak base.
Is nh4i an acid or base?
Ammonium iodide is an acid because when it dissolves in water, some of the ammonium ions will react with water forming ammonia (NH3) and hydronium,…
Is aluminum nitrate a strong electrolyte?
The compound aluminum nitrate is a strong electrolyte.
Is KNO2 an acid or base?
Potassium nitrite, KNO2, has to be a basic salt. Potassium nitrite is a salt of a strong base (sodium hydroxide, NaOH) and a weak acid (nitrous acid, HNO2). In aqueous solution such a salt undergoes anionic hydrolysis to a certain extend and produces hydroxide ions (OH-) as per the following equilibrium.Is NO3 3 weak or strong?
Aluminium hydroxide is a weak base but nitric acid is a strong acid. The salt is formed by reacting strong acid and weak base is known as acidic salt. Since aluminum nitrate(salt),Al(NO3)3 is formed by reacting strong acid,HNO3 and weak base,Al(OH)3, its acidic in nature.
Is NH4I a strong or weak acid?
NH4I is a salt of hydroiodic acid (HI), a strong acid, and ammonia (NH3), a weak base. An aqueous solution of such a salt is acidic (pH < 7 ) due to hydrolysis of the cation.
Is NO3 a strong base?
NO3- is derived from HNO3, one of the strong acids, so this ion is also neutral. The nitrate anion NO3- is the weak conjugate base of the strong acid HNO3.
Is Fe NO3 3 an acid or base?
Fe(NO3)3 —This salt was formed from the reaction of a weak base, iron (III) hydroxide, with a strong acid, nitric acid. This means that the salt will be acidic.Is NH4I an ionic compound?
Ammonium iodide is an ionic compound, which means that both ammonium and iodine have a charge. Ammonium…
Is KNO2 strong acid?Therefore, neither ion will affect the acidity of the solution, so KCl is a neutral salt. Although the K + ion derives from a strong base (KOH), the NO 2 − ion derives from a weak acid (HNO 2). Therefore the solution will be basic, and KNO 2 is a basic salt.
Article first time published onIs KNO3 a strong acid?
Name of MoleculePotassium nitratepH7
Is formic acid a strong electrolyte?
Formic acid, HCOOH, is a weak electrolyte.
Is aluminum a strong electrolyte?
The compound aluminum chloride is a strong electrolyte.
Does Al no3 3 undergo hydrolysis?
The pH of the Aluminum Nitrate Solution: The weak acid/base ion will then undergo hydrolysis in a solution that affects the pH of the solution.
What ion is no3?
ChEBI NamenitrateDefinitionA nitrogen oxoanion formed by loss of a proton from nitric acid. Principal species present at pH 7.3.
Which are strong acids and bases?
Strong AcidsStrong Baseshydrobromic acid (HBr)potassium hydroxide (KOH)hydroiodic acid (Hl)calcium hydroxide (Ca(OH)2)nitric acid (HNO3)strontium hydroxide (Sr(OH)2)sulfuric acid (H2SO4)barium hydroxide (Ba(OH)2)
Is Ethanoic acid a strong acid?
Strong acids dissociate fully in water to produce the maximum number of H + ions. … Weak acids, such as ethanoic acid (CH 3COOH), do not fully dissociate.
Is phosphoric acid a strong acid?
AcidHCN (hydrocyanic acid) (weakest)CN− (cyanide ion) (strongest)
Is no3 an acid or base in water?
KaAcidBase2.4 * 101Nitric acidNitrate ion——–Hydronium ionWater5.4 * 10-2Oxalic acidHydrogen oxalate ion1.3 * 10-2Sulfurous acidHydrogen sulfite ion
Does NH4I undergo hydrolysis?
When ammonium iodide dissolves in water it dissociates into ammonium and iodide. The iodide ion does not undergo hydrolysis, but ammonium undergoes…
Is carbonate an anion or cation?
Property NameProperty ValueReferenceFormal Charge-2Computed by PubChem
Is Fe OH 3 an acid?
All other Arrhenius acids and bases are weak acids and bases. For example, acetic acid (HC2H3O2) and oxalic acid (H2C2O4) are weak acids, while iron hydroxide, Fe(OH)3, and ammonium hydroxide, NH4OH (which is actually just ammonia, NH3, dissolved in water), are examples of weak bases.
Is HBr a strong acid?
Hydrobromic acid is a strong acid formed by dissolving the diatomic molecule hydrogen bromide (HBr) in water. … Hydrobromic acid is one of the strongest mineral acids known.
Is Fe acidic?
1. Fe(NO3)3 —This salt was formed from the reaction of a weak base, iron (III) hydroxide, with a strong acid, nitric acid. This means that the salt will be acidic.
Is H2CO3 a strong acid?
H2CO3 is a strong acid.
Is HBr a strong base?
AcidsBasesHBrNaOHHIKOHHNO 3RbOHH 2SO 4CsOH
What is the basicity of nitrous acid?
Monobasic: Since Nitrous acid HNO2 has only one hydrogen ion which it can donate to a base in case of a reaction between an acid and a base, it is known as a monobasic acid. It can release only one proton, i.e., H+ in solution.
What is ionic compound KNO2?
Potassium nitrite | KNO2 – PubChem.
What type of salt is KNO2?
Potassium nitrite (distinct from potassium nitrate) is the inorganic compound with the chemical formula KNO 2. It is an ionic salt of potassium ions K+ and nitrite ions NO2−, which forms a white or slightly yellow, hygroscopic crystalline powder that is soluble in water.
Is Na2SO4 a strong acid?
The salts produced from combining strong acids and bases (NaCl, NaNO3, Na2SO4, KCl, KNO3, K2SO4) are neutral salts. If these neutral salts are dissolved in water, the solution pH is close to 7.0. Strong acids react with weak bases, such as ammonia (ammonium hydroxide).
Is KNO3 a solid?
Potassium Nitrate is a transparent, white or colorless, crystalline (sand-like) powder or solid with a sharp, salty taste. It is used to make explosives, matches, fertilizer, fireworks, glass and rocket fuel.