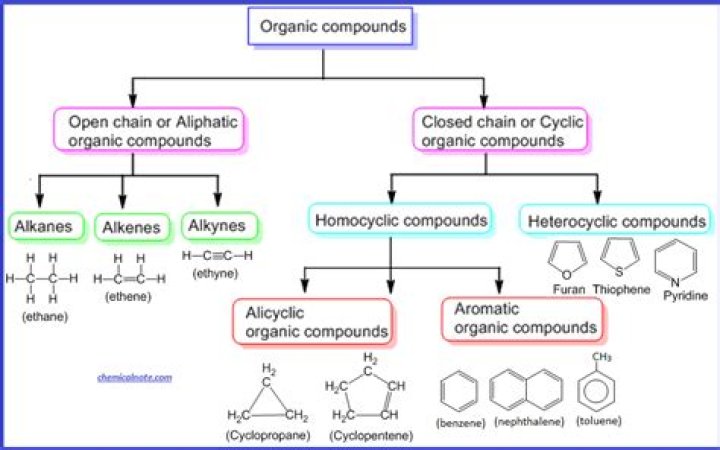
The organic acids are categorized based on four characterizations: (1) the nature of carbon chain (aromatic, aliphatic, alicyclic, and heterocyclic); (2) saturation or unsaturation properties; (3) substituted or nonsubstituted features; and (4) the number of functional groups (mono, di- or tri-carboxylic).
What are the 5 steps to naming an organic compound?
- Step 1: Locate the longest carbon chain in our compound. …
- Step 2: Name that longest carbon chain. …
- Step 3: Figure out what the ending (suffix) should be. …
- Step 4: Number your carbon atoms. …
- Step 5: Name the side groups. …
- Step 6: Put the side groups in alphabetical order.
What do you understand by organic acids give the name of the organic acids and their sources?
An organic acid is an organic compound with acidic properties. The most common organic acids are the carboxylic acids, whose acidity is associated with their carboxyl group –COOH. Sulfonic acids, containing the group –SO2OH, are relatively are stronger acids.
How do you identify an acid or base in organic chemistry?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).How do you name organic?
- Find and name the longest continuous carbon chain.
- Identify and name groups attached to this chain.
- Number the chain consecutively, starting at the end nearest a substituent group.
- Designate the location of each substituent group by an appropriate number and name.
How do you name things in organic chemistry?
- Prefix = substituent.
- First Name = carbon chain number.
- Last Name = type of chain.
- Suffix = highest priority functional group.
How do you name organic molecules?
In summary, the name of the compound is written out with the substituents in alphabetical order followed by the base name (derived from the number of carbons in the parent chain). Commas are used between numbers and dashes are used between letters and numbers. There are no spaces in the name.
How can you identify an acid or base without litmus paper?
In the absence of litmus paper, a reagent phenolphthalein can be used. The solution which develops pink colour with phenolphthalein is a base and the other is acid.What is an acid in organic chemistry?
An organic acid is an organic compound with acidic properties. The most common organic acids are the carboxylic acids, whose acidity is associated with their carboxyl group –COOH. … The relative stability of the conjugate base of the acid determines its acidity.
How do you know if an acid or base is pH?A solution’s pH will be a number between 0 and 14. A solution with a pH of 7 is classified as neutral. If the pH is lower than 7, the solution is acidic. When pH is higher than 7, the solution is basic.
Article first time published onWhich one of the given is an organic acid Mcq?
(D) Nitric acid Mineral acids are the ones which are derived from inorganic compounds. Hydrochloric acid, sulphuric acid and nitric acid are mineral acids whereas citric acid is an organic acid.
What are acids name a few naturally occurring acids and their sources?
- Vinegar: Acetic acid.
- Apple: Malic acid.
- Tamarind: Tartaric acid.
- Milk: Lactic acid.
- Lemon: Citric acid.
- Tomato: Ascorbic acid.
- Grapes: Tartaric acid.
How are organic acids different from mineral acids?
The key difference between mineral acids and organic acids is that the mineral acids do not essentially contain carbon and hydrogen whereas organic acids essentially contain carbon and hydrogen atoms. … Hence, they are organic compounds having acidic properties.
How do you name hydrocarbons?
1. The first part of the name is based on the length of the longest carbon chain in the molecule. 2. The end of the name is given by the number of bonds between carbon atoms.
How do you name hydrocarbons step by step?
- STEP 1 Find the root: Identify the longest chain or ring in the hydrocarbon. …
- STEP 2 Find the suffix: If the hydrocarbon is an alkane, use the suffix –ane. …
- STEP 3 Give a position to every atom in the main chain. …
- Step 5 Put the name together: prefix + root + Suffix.
How do you name carboxylic acids?
In general, carboxylic acids are named based on the number of carbons in the longest continuous chain, including the carboxyl group (-COOH). The suffix of this carbon chain is then replaced, as carboxylic acids always end in “-oic acid.” An example is CH2O2, in which the longest continuous carbon chain is a methane.
How do you name organic compounds Class 11?
Class of Organic compoundFunctional GroupSecondary Suffixcarboxylic acid-COOH-oic acidAcid amides-CONH2-amideAcid chloride-COCl-oyl chlorideEsters-COORalkyl oate
How do you name organic compounds with multiple functional groups?
Organic Chemistry Here is the trick – you need to identify the functional group with the highest priority and add a suffix (ending) of that functional group. The other groups are treated as substituents and added to the name with prefixes: Let’s break this down to see how it works.
How do you name alkanes?
- The locant: The number indicating where the substituent is.
- The prefix: The substituent attached to the alkane. Ends with -yl.
- The Parent: The alkane parent chain. Ends with -ane.
- Suffix: The functional group attached to the alkane. Not always present.
How do you name polymers in organic chemistry?
To name an addition polymer with only one monomer, you insert the name of the monomer in parentheses after the “poly” prefix: for example, “poly(methyl methacrylate).” If the name could indicate several different compounds, the class of polymer can be used to clarify, such as “polyether.” If the monomer is one word …
How do you name alkanes with branches?
- Count the longest continuous chain of carbons. …
- Number the carbons in the chain starting with the end that’s closest to a branch. …
- Count the number of carbons in each branch. …
- Attach the number of the carbon from which each substituent branches to the front of the alkyl group name.
What are organic acids Class 10?
Acids are those substances which are sour in taste. … The acids present in plant materials and animals are called organic acids. For Example:Acetic acid (Vinegar), Citric acid (lemon, orange), Lactic acid (curd or milk), Tartaric Acid (tamarind), Formic acid etc.
How do you make organic acid?
Generally, organic acids are produced commercially either by chemical synthesis or fermentation. However, fermentation processes are the most commonly used method. All organic acids of tricarboxylic acid cycle can be produced in high yields in microbiological processes.
What is the example of organic acid?
Examples of organic acids include tartaric acid and formic acid.
What is the best way to differentiate an acid and a base?
Acid is a kind of chemical compound that when dissolved in water gives a solution with H+ ion activity more than purified water. A base is an aqueous substance that donates electrons, accept protons or release hydroxide (OH-) ions.
How do we test for acids?
- Litmus.
- Methyl orange.
- Phenolphthalein.
How can u tell if something is an acid?
The use of the pH scale is one practical way of quantitatively determining how acidic something is. If the pH of a solution is less than 7, it is acidic. If the pH is 7, the solution is neutral and if the pH is greater than 7,the solution is basic.
Is white vinegar a base or acid?
Vinegar is acidic. Vinegar’s pH level varies based upon the type of vinegar it is. White distilled vinegar, the kind best suited for household cleaning, typically has a pH of around 2.5.
What does pH stand for?
pH may look like it belongs on the periodic table of elements, but it’s actually a unit of measurement. The abbreviation pH stands for potential hydrogen, and it tells us how much hydrogen is in liquids—and how active the hydrogen ion is.
How do you find ka?
As noted above, [H3O+] = 10-pH. Since x = [H3O+] and you know the pH of the solution, you can write x = 10-2.4. It is now possible to find a numerical value for Ka. Ka = (10-2.4)2 /(0.9 – 10-2.4) = 1.8 x 10-5.
Which one of the given is an organic acid answer?
One of the most common organic acids is carboxylic acid, which has the molecular formula RCOOH. Other common organic acids include sulfonic acid and alcohol.